reading is too low. A hydrometer reading is
inaccurate if you take it immediately after water
is added. This is because the water tends to
remain at the top of the cell. When water is added,
charge the battery for at least an hour. This will
mix the electrolyte before the hydrometer reading
is taken.
CAUTION
After use, flush hydrometers with fresh
water. This prevents inaccurate readings.
Do NOT use storage battery hydrometers
for any other purpose.
Correcting Specific Gravity
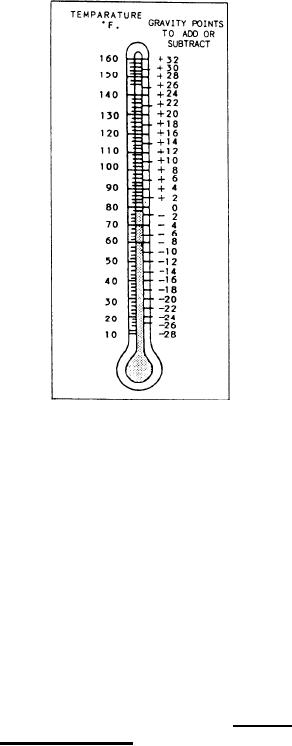
Temperature affects the specific gravity of
electrolyte. When electrolyte is heated, it expands
and becomes less dense. Its specific gravity reading
is lowered. On the other hand, when electrolyte
is cooled, it contracts and becomes denser when
cooled. Its specific gravity reading is raised. In
Figure 10-11 .--Temperature conversion chart.
both cases, the electrolyte may be from the same
fully charged storage cell.
When measuring the specific gravity, you must
weight, or about 27 percent by volume. Distilled
also measure the temperature of the electrolyte.
water and sulfuric acid are used to prepare the
Most standard storage batteries use 80F as the
electrolyte. New batteries may be delivered with
normal temperature to which specific gravity
containers of concentrated sulfuric acid of 1.830
readings are corrected. Figure 10-11 shows a
specific gravity or electrolyte of 1.400 specific
temperature conversion chart for an 80F
gravity. You must dilute both of these with
hydrometer. You can also correct the specific
distilled water to make electrolyte of the proper
gravity reading of a storage battery by adding 1
specific gravity. Use a container made of glass,
point for each 3F above 80F, or by subtracting
earthenware, rubber, or lead for diluting the acid.
1 point for each 3F below 80F.
Adjusting Specific Gravity
WARNING
Sometimes the specific gravity of a cell is more
When mixing electrolyte, ALWAYS
than it should be. You can reduce it to within
POUR ACID INTO WATER--NEVER
limits by removing some of the electrolyte and
pour water into acid. Pour the acid slowly
adding distilled water. As stated earlier, charge
and cautiously to prevent excessive heating
the battery for 1 hour to mix the solution. Then
and splashing. Stir the solution con-
take hydrometer readings. Continue the adjust-
tinuously with a nonmetallic rod to mix the
ment until you get the desired true readings.
heavier acid with the lighter water. This
will keep the acid from sinking to the
NOTE: Only authorized personnel should add
bottom.- When concentrated -acid is
diluted, the solution becomes very hot.
acid to a battery. Acid with a specific gravity
above 1.350 is NEVER added to a battery.
TREATMENT OF SPILLED ACID
MIXING ELECTROLYTE
If acid or electrolyte from a lead-acid battery
The electrolyte of a fully charged battery
comes into contact with the skin, wash the
usually has about 38 percent sulfuric acid by
affected area with a large amount of fresh water.
10-13



