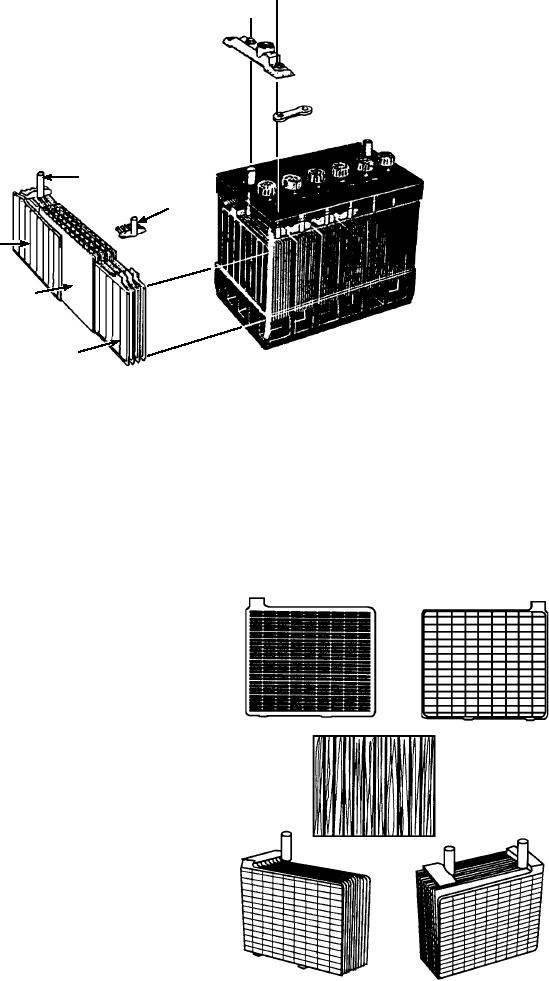
HARD RUBBER
CELL COVER
CELL CONNECTOR
TERMINAL POST
PLATE
STRAP
NEGATIVE
CASTING
PLATE
GROUP
SEPARATOR
POSITIVE
PLATE
GROUP
ASf06008
Figure 6-8.--Battery construction.
in series to form a battery, it makes a noticeable
BATTERIES
difference in the battery voltage (6.45 volts for the
LEARNING OBJECTIVES: Identify the
6-volt battery, 12.9 volts for the 12-volt battery, and
types of SE batteries. Identify procedures for
25.8 volts for the 24-volt battery). For purposes of
inspecting, testing, and servicing SE batteries.
discussion, however, we will refer to the cells as having
Identify the procedures for troubleshooting SE
a voltage of 2.0 volts.
batteries. Identify the procedures for removing
Each cell contains positive lead plates and negative
and replacing SE batteries.
lead plates. As shown in figures 6-8 and 6-9, these
The most commonly used batteries in support
equipment are lead-acid batteries. The battery is used to
start the engine, and it aids the generator in meeting the
electrical system's power demands under low rpm and
heavy load conditions. The battery can also supply a
limited amount of power for a short time without
operating the generator. Active materials within the
battery react chemically to produce a flow of direct
PLATE GRID
PLATE
current whenever a load is connected. This current is
produced by chemical reaction between the active
materials of the lead plates and the sulfuric acid of the
electrolyte.
CONSTRUCTION
WOOD SEPARATOR
A battery consists of a number of cells connected
together. The number of cells depends on the voltage
desired. Three cells connected in series make up what is
commonly called a 6-volt battery. A 12-volt battery is
made up of six cells connected in series, and a 24-volt
battery has 12 cells connected in series. Most
references on batteries state that each cell has a voltage
PLATE GROUP
ELEMENTS
of 2.0 volts. But each cell actually produces about 2.15
ASf06009
volts (no load) if a very accurate voltmeter is used and
the cell is fully charged. When the cells are connected
Figure 6-9.--Cell element construction.
6-9

